For producing lentiviral particles you typically need three components:
- 1) a lentiviral vector, such as pLKO.1 or pLVTHM or pLVX-PURO, containing the shRNA or transgene,
- 2) a packaging vector, such as psPAX2 or pCMV-dR8.2 dvpr and
- 3) an envelop vector, such as pMD2.G or pCMV-VSVG.
For most applications, you can produce viral particles by transient transfection of 293T cells with a 2nd generation packaging system(e.g.: packaging plasmid psPAX2 and envelop plasmid pMD2.G).

Fig 1. Viral Production Schematic (from addgene.org)
Lentiviral vector production systems have been refined over time to improve their performance and safety. Depending on their features, vector systems are divided into different generations. The higher the generation, the safer the vector.
First-generation
The first-generation lentiviral vectors were manufactured using a packaging system that comprised all HIV genes, except the env gene (usually heterologous), which is separated in one plasmid. This system consists of 3 plasmids:
- 1) Transfer vector;
- 2) Packaging plasmid expressing gag, pol, tat, rev and accessory genes;
- 3) Envelop plasmid expressing env gene.
Second-generation
It was shown subsequently that none of the four HIV-1 accessory genes vif, vpr, vpu, or nef were required for HIV-1 replication in immortalized cell lines. This led to the development of a “second generation” of HIV-1 vector systems.
In this system, the accessory genes were eliminated leaving the gag and pol reading frames, which encode for the structural and enzymatic components of the virion, respectively, and the tat and rev genes, fulfilling transcriptional and post-transcriptional functions.In general, lentiviral vectors with a wild type 5’ LTR need the 2nd generation packaging system because these vectors require tat for activation. In this system, the packaging plasmid expresses gag, pol, tat and rev. All lentiviral vectors from the Trono or Aebischer lab require packaging with a 2nd generation system.
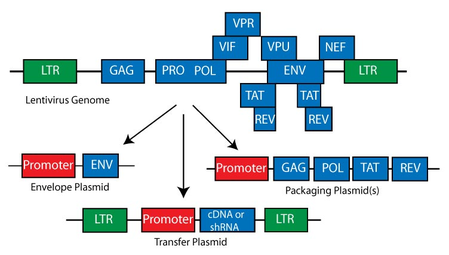
Fig 2. Second Generation Lentiviral Plasmids (from addgene.org)
2nd generation system deposited by the Trono lab:
psPAX2: 2nd generation packaging plasmid for producing viral particles. psPAX2 contains a robust CAG promoter for efficient expression of packaging proteins. Trono lab and Aebischer lab lentiviral vectors require psPAX2. Produces higher titer than pCMV-dR8.2 dvpr.
pMD2.G: Envelop plasmid for producing viral particles.
2nd generation system deposited by the Weinberg lab:
pCMV-dR8.2 dvpr: 2nd generation packaging plasmid for producing viral particles.
pCMV-VSVG: Envelop plasmid for producing viral particles.
2nd generation Lentiviral Transfer Plasmids
Third-generation
In a third-generation system, only gag, pol, and rev genes remain present (tat is eliminated). The rev gene is provided in a separated plasmid. Since the HIV promoter in 5’ LTR depends on tat, a vector which lacks tat needs to have its wild type promoter replaced with a heterologous enhancer/promoter such as CMV or RSV to ensure transcription. The HIV-1 Tat protein interacts with TAR RNA and recruits CDK9/cyclin T1 and other host factors to induce HIV-1 transcription.
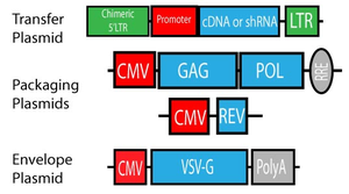
Fig 3. Third Generation Lentiviral Plasmids (from addgene.org)
The 3rd generation packaging system offers maimal biosafety but is more cumbersome to user, as it involves the transfection of four different plasmids in the producer cells.
If you wish to use this system, you need to have a lentiviral vector with a chimeraic 5' LTR in which the HIV promoter is replaced with CMV or RSV, thus making it TAT-independent. Examples of these vectors include pLKO.1, pLL3.7, pLB, pLenti6, pSico, pCL, pCDH and pCS. Most Aebischer and Trono lab lentiviral vectors CANNOT be used with this system.
A transfer vector carrying a chimeric 5' LTR can be packaged with either the 2nd or 3rd generation packaging system. A 3rd generation transfer plasmid can be used with a 2nd generation packaging system, but a 2nd generation transfer plasmid cannot be used with a 3rd generation packaging system.
3rd generation system plasmids
pMDLg/pRRE: 3rd generation packaging plasmid for producing viral particles.
pRSV-Rev: 3rd generation packaging plasmid for producing viral particles.
pMD2.G:Envelop plasmid for producing viral particles.
3rd generation Lentiviral Transfer Plasmids
Souce: NovoPro 2021-12-13
