-
Product Name
Anti-Hsp90 alpha (2H2) Mouse antibody
- Documents
-
Description
Hsp90 alpha (2H2) Mouse monoclonal antibody
-
Tested applications
WB, IHC-P
-
Species reactivity
Human, Rat, Mouse
-
Isotype
Mouse IgG1
-
Preparation
Antigen: Purified recombinant protein expressed in E.coli.
-
Clonality
Monoclonal
-
Formulation
PBS(pH 7.4) containing with 0.02% sodium azide and 50% glycerol.
-
Storage instructions
Store at 4°C short term. Store at -20°C long term. Avoid freeze / thaw cycle.
-
Applications
WB: 1/2;000-5;000
IHC: 1/100-200
-
Validations
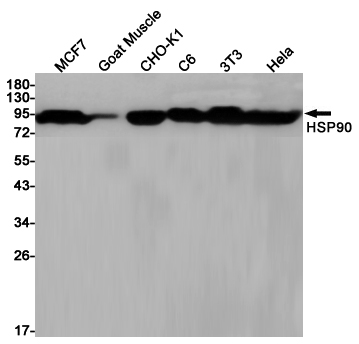
Western blot detection of HSP90 in MCF7,Goat Muscle,CHO-K1,C6,3T3,Hela cell lysates using HSP90 (2H2) Mouse mAb(1:1000 diluted).Predicted band size:95KDa.Observed band size:90KDa.
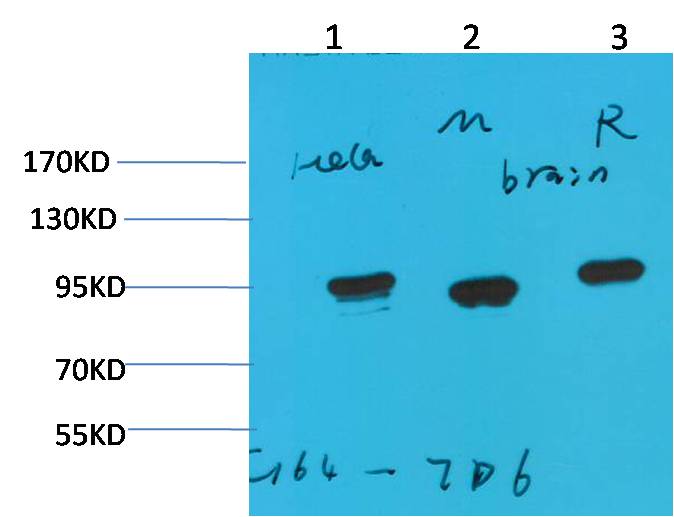
Western blot analysis of 1)Hela, 2)Mouse Brain Tissue, 3) Rat Brain Tissue with HSP90 a Mouse mAb diluted at 1:2,000.
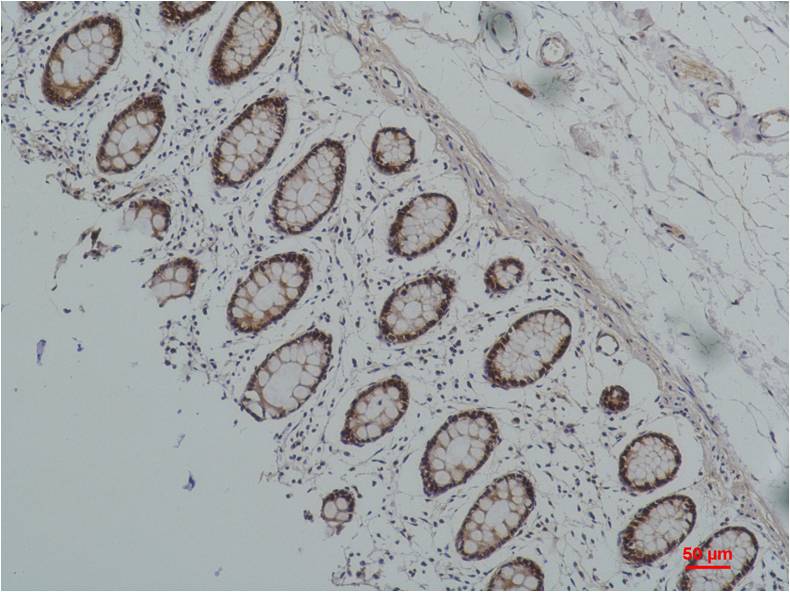
Immunohistochemical analysis of paraffin-embedded Human Colon Caricnoma using HSP90 alpha; Mouse mAb diluted at 1:200.
-
Background
Swiss-Prot Acc.P07900.Molecular chaperone that promotes the maturation, structural maintenance and proper regulation of specific target proteins involved for instance in cell cycle control and signal transduction. Undergoes a functional cycle that is linked to its ATPase activity which is essential for its chaperone activity. This cycle probably induces conformational changes in the client proteins, thereby causing their activation. Interacts dynamically with various co-chaperones that modulate its substrate recognition, ATPase cycle and chaperone function (PubMed:11274138, PubMed:15577939, PubMed:15937123, PubMed:27353360). Engages with a range of client protein classes via its interaction with various co-chaperone proteins or complexes, that act as adapters, simultaneously able to interact with the specific client and the central chaperone itself. Recruitment of ATP and co-chaperone followed by client protein forms a functional chaperone. After the completion of the chaperoning process, properly folded client protein and co-chaperone leave HSP90 in an ADP-bound partially open conformation and finally, ADP is released from HSP90 which acquires an open conformation for the next cycle (PubMed:27295069, PubMed:26991466). Apart from its chaperone activity, it also plays a role in the regulation of the transcription machinery. HSP90 and its co-chaperones modulate transcription at least at three different levels. In the first place, they alter the steady-state levels of certain transcription factors in response to various physiological cues. Second, they modulate the activity of certain epigenetic modifiers, such as histone deacetylases or DNA methyl transferases, and thereby respond to the change in the environment. Third, they participate in the eviction of histones from the promoter region of certain genes and thereby turn on gene expression (PubMed:25973397). Binds bacterial lipopolysaccharide (LPS) and mediates LPS-induced inflammatory response, including TNF secretion by monocytes (PubMed:11276205). Antagonizes STUB1-mediated inhibition of TGF-beta signaling via inhibition of STUB1-mediated SMAD3 ubiquitination and degradation (PubMed:24613385).
Related Products / Services
Please note: All products are "FOR RESEARCH USE ONLY AND ARE NOT INTENDED FOR DIAGNOSTIC OR THERAPEUTIC USE"
