-
Product Name
Anti-Cpn10 Rabbit antibody
- Documents
-
Description
Cpn10 Rabbit polyclonal antibody
-
Tested applications
WB, IHC-P, IP
-
Species reactivity
Human, Mouse, Rat
-
Alternative names
EPF; CPN10; GROES; HSP10 antibody
-
Isotype
Rabbit IgG
-
Preparation
Antigen: A synthetic peptide of human Cpn10
-
Clonality
Polyclonal
-
Formulation
Supplied in 50nM Tris-Glycine(pH 7.4), 0.15M Nacl, 40%Glycerol, 0.01% sodium azide and 0.05% BSA.
-
Storage instructions
Store at -20°C. Stable for 12 months from date of receipt.
-
Applications
WB: 1/2000-1/10000
IHC: 1/50-1/200
IP: 1/20-1/50
-
Validations

Western blot detection of Cpn10 in K562,Hela cell lysates using Cpn10 Rabbit pAb(1:1000 diluted).Predicted band size:11kDa.Observed band size:11kDa.
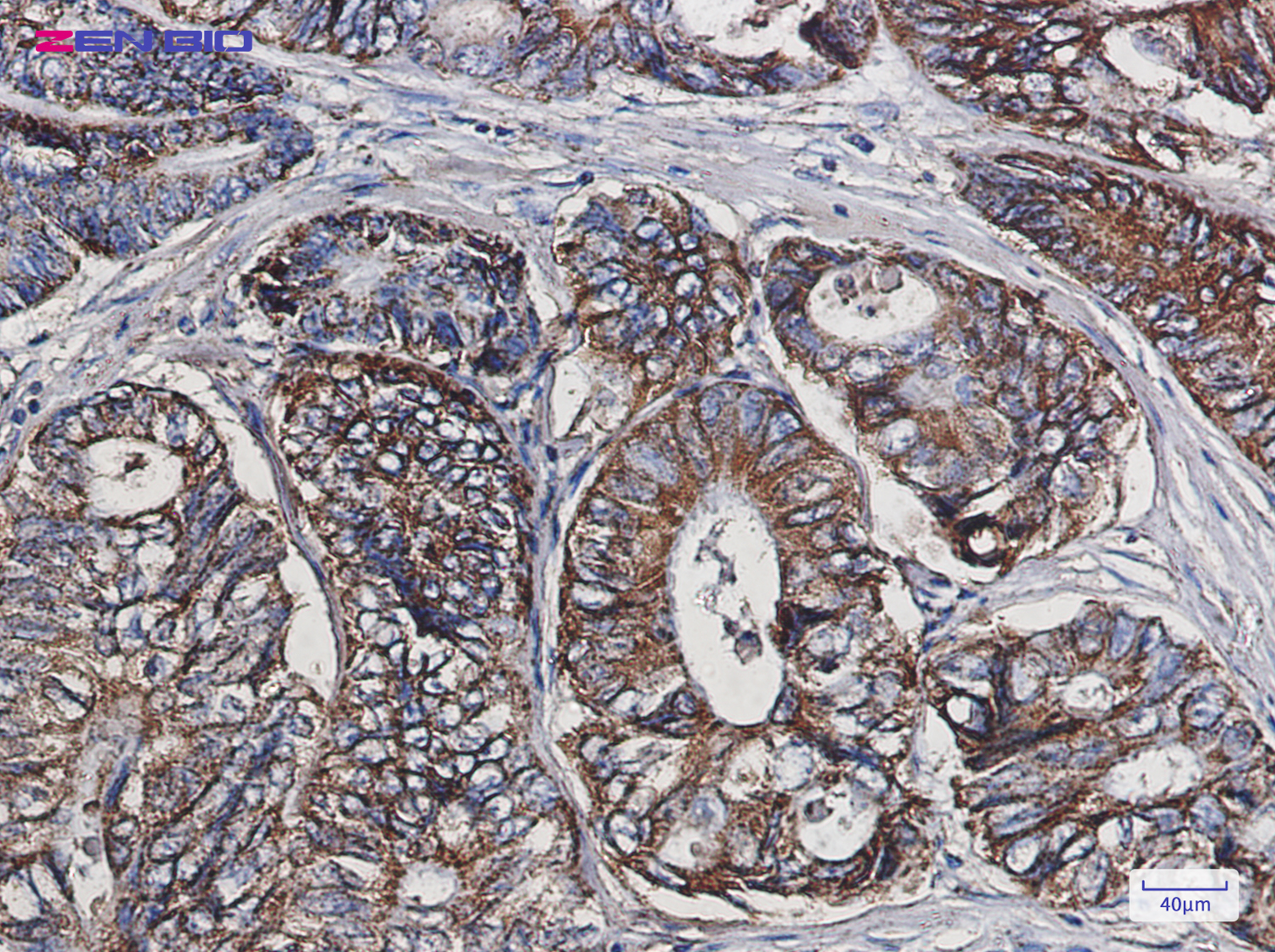
Immunohistochemistry of Cpn10 in paraffin-embedded Human colon cancer tissue using Cpn10 Rabbit pAb at dilution 1/50
-
Background
Swiss-Prot Acc.P61604.Co-chaperonin implicated in mitochondrial protein import and macromolecular assembly. Together with Hsp60, facilitates the correct folding of imported proteins. May also prevent misfolding and promote the refolding and proper assembly of unfolded polypeptides generated under stress conditions in the mitochondrial matrix (PubMed:7912672, PubMed:1346131, PubMed:11422376). The functional units of these chaperonins consist of heptameric rings of the large subunit Hsp60, which function as a back-to-back double ring. In a cyclic reaction, Hsp60 ring complexes bind one unfolded substrate protein per ring, followed by the binding of ATP and association with 2 heptameric rings of the co-chaperonin Hsp10. This leads to sequestration of the substrate protein in the inner cavity of Hsp60 where, for a certain period of time, it can fold undisturbed by other cell components. Synchronous hydrolysis of ATP in all Hsp60 subunits results in the dissociation of the chaperonin rings and the release of ADP and the folded substrate protein (Probable).
Related Products / Services
Please note: All products are "FOR RESEARCH USE ONLY AND ARE NOT INTENDED FOR DIAGNOSTIC OR THERAPEUTIC USE"
