-
Product Name
Human alpha-2-macroglobulin (His Tag) recombinant protein
- Documents
-
Description
Is able to inhibit all four classes of proteinases by a unique 'trapping' mechanism. This protein has a peptide stretch, called the 'bait region' which contains specific cleavage sites for different proteinases. When a proteinase cleaves the bait region, a conformational change is induced in the protein which traps the proteinase. The entrapped enzyme remains active against low molecular weight substrates (activity against high molecular weight substrates is greatly reduced). Following cleavage in the bait region a thioester bond is hydrolyzed and mediates the covalent binding of the protein to the proteinase.
-
Protein name
Alpha-2-macroglobulin
-
Protein short names
A2MD; S863-7; CPAMD5; A2M; FWP007; ALPHA-2-MACROGLOBULIN; A2MP; DKFZP779B086
-
Uniprot ID
P01023
-
Gene Name
A2M; CPAMD5; FWP007
-
Source/Expression Host
Baculovirus-Insect Cells
-
Expression Plasmid/cDNA
A DNA sequence encoding the human A2M (NP_000005.2) (Met 1-Ala 1474) was expressed, fused with a polyhistidine tag at the C-terminus.
-
Protein Species
Human
-
Molecular weight
The recombinant human A2M consists of 1461 amino acids and predicts a molecular mass of 164 kDa. The apparent molecular mass of rhA2M is approximately 160-170 kDa in SDS-PAGE under reducing conditions.
-
Purity
> 92 % as determined by SDS-PAGE
-
Activity
Measured by its ability to trap trypsin. The trapped trypsin is no longer able to interact with protein substrates or inhibitors, but still able to cleave small peptide substrates or inhibitors.
The IC50 value is <5 nM. -
Validations
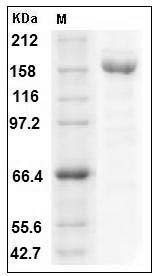
Human A2M / CPAMD5 / Alpha-2-macroglobulin Protein (His Tag) SDS-PAGE
Related Products / Services
Please note: All products are "FOR RESEARCH USE ONLY AND ARE NOT INTENDED FOR DIAGNOSTIC OR THERAPEUTIC USE"
